Philips IntelliSite Pathology Solution has market clearance in EEA (European Economic Area), United Kingdom, Ireland and Singapore. Specific conditions apply to the USA market.1
Meet our Philips team at ECP 2023
Sept 9-12, Convention Centre Dublin, Ireland
Discover the full potential of digital pathology with our expert team! We will showcase how we can help you see beyond digital pathology and unlock the power of integrated diagnostics. Join us at our booth #43 and let's take the first step towards your digital transformation, together.
Philips IntelliSite Pathology Solution
Introducing Philips IntelliSite Pathology Solution: the next generation of digital pathology products, designed to let you review and collaborate anytime possible. Philips offers tools and resources for every stage of the pathology process, for a comprehensive, end-to-end experience that allows you to: Gone are the days of tedious microscope viewing, inefficient slide storage, and crucial clinical decisions made in silos. Instead, experience a digital workflow that empowers your diagnosticians by eliminating inefficiencies and enabling collaboration like never before – to unlock the full possibilities of precision medicine.
High quality digitization that truly enables digital pathology networks
The Pathology Scanner Second Generation family will address current unmet market needs for networked pathology for primary diagnosis and is able to provide a fully dedicated solution for small, medium and large volume laboratories based on their workflow needs. The Pathology Second Generation Scanner family consist of three high performing scanner models with different slide capacities using the same scan engine across the models delivering consistently high image quality and first-time right rates, resulting in a very low cost of operation and / or low total cost of ownership (TCO) per slide. Furthermore, the solution with new 3-D ready technologies incorporated, and hardware that is prepared for multi-layer scanning.
-
Pathology Scanner SG60
SG60 is designed to accommodate laboratories with a lean workflow and need to scan small batches of slides to achieve operational excellence and short turnaround times by scanning batches in parallel. With a high throughput, high first time right rate and load and walk away scanning, the SG60 enables you to digitize your histology samples and obtain high quality clinical diagnostic images for routine use and integrated pathology networks.
FDP0909 -
Pathology Scanner SG300
SG300 is designed to accommodate laboratories for high volume labs that want to maximize scanner utilization and further reduce the total cost of ownership per slide by means of overnight scanning. With a high throughput, high first time right rate and load and walk away scanning, the SG300 enables you to digitize your histology samples and obtain high quality clinical diagnostic images for routine use and integrated pathology networks.
FDP0911
Demonstrated technology in time savings and throughput performance*
96%

'I experience an increase in my productivity for routine diagnosis with PIPS'
100%

'Philips delivers a platform which is reliable as a microscope when doing diagnosis'
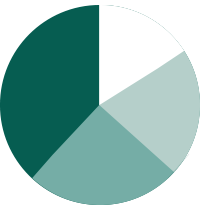

>10%

>20%

>15%

>25%
'What is your productivity gain due to Philips IntelliSite Pathology Solutions'

Enhanced patient care

Reduced costs due to efficiency and productivity gains
Improved workflow compared to analog

A proven solution that scales as you grow

The IMS case view simplifies access to histopathology cases
* Philips DCP research study, results are specific to the institution where they were obtained and may not reflect the results achievable at other institutions (2018)
On demand webinar
Digitization of pathology: step towards connected care and precision medicine.

MD, PhD
Medical Officer - Oncology Philips
Global
On demand webinar
How to successfully leave the microscope behind, diagnostic workflows and the mindset of the pathologists

Dr. Charlotte Orndal, MD Unilabs Pathology Sweden
On demand webinar
First large scale multicenter digital pathology implementation in Finland - project Fimlab

Dr. Teemu Tolonen, MD, PhD Fimbab Laboratories Finland
Customer references
1The expansion of remote use of Philips IntelliSite Pathology Solution in the USA are under specific conditions and device specifications. The type of monitors is only allowed due to emergency situations. The limitations and warnings specified in device labeling are still applicable to this situation 2Survey of 52 pathologists, lab managers and lab technicians in Europe, 2018. Results are specific to the institution where they were obtained and may not reflect the results achievable at other institutions.


